  
In an Atom, Electrons have discrete and some specific energies. Each element has a characteristic Spectrum through which it can easily be recognized. I'm studying emission spectrums at school right now, and my teacher said we're going to do a "flame test" for our astronomy course next week, but I have no idea what that is. The Atomic spectra are defined as the Spectrum of frequencies of electromagnetic radiation emitted or absorbed during transitions of Electrons between Energy levels within an Atom. In an emission spectra electrons are excited to an excited state by thermal or electrical means and then relax back to a lower state and emit a photon of light at a specific energy, which is seen as at a specific wavelength. Your instructor must have mentioned to you about Bunson and Kirchhoff who first studied the atomic emission spectrum by using a burner flame and a prism. Line Spectra There are two types of line spectra, emission and absorption. The discontinuous line spectra of light produced when excited atoms return to their ground state and emit photons of a certain. protons and neutrons in nucleus, orbit represented by n, each energy level holds a max of 2n2. improved from rutherford, electrons orbit nucleus on fixed path. How it works is that when an atom or molecule is heated, it emits radiation which can be seen as a spectrum of bright lines. each line (color) corresponds to a specific relaxation of an electron from a higher energy level to a lower energy level. Because if an x-ray is an emission spectrum, then why aren't we able to see it?įebruary 4, A flame test is a test where an element is literally held to a flame so that it emits a spectrum of color and by that color, you can determine what that element is. Hence, a large molecular band spectrum is observed. I had heard that an x-ray is also an emission spectrum, but after reading this I'm kind of doubting it. Study with Quizlet and memorize flashcards containing terms like The emission or absorption spectrum for calcium in a low-temperature flame is broad because the calcium is largely present as the molecule CaOH, which has many vibrational and rotational states and thus many excited energy levels. We can see sunlight because it has the right wavelength, but we can't see radio waves. an X-ray's wavelength is too short for us to see. We're not able to see x-rays because not all spectrum is visible. He observes two discrete lines at 600 nm and. An emission line will appear in a spectrum if the source emits specific wavelengths of radiation. Tyrone analyzes the emission spectrum of an unknown element. 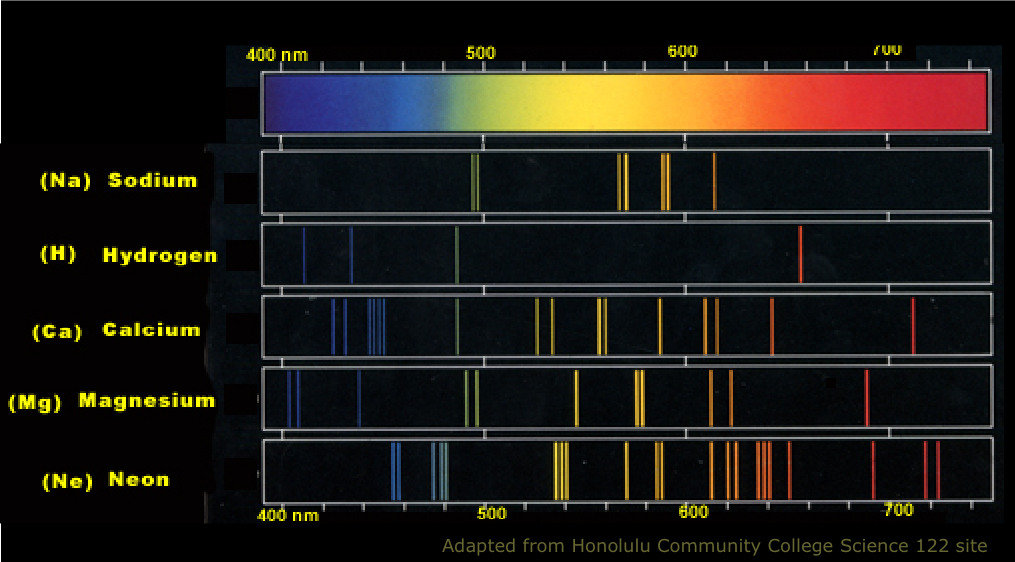
So you can think of x-rays as a single piece that makes up an emission spectrum. A table of the visible light spectrum is shown below. When electromagnetic radiation comes together in an arrangement, its called an electromagnetic spectrum. February 8, Actually an x-ray is not an emission spectrum, its electromagnetic radiation. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
